| Applications such as electric vehicles and renewable energy sources require the development of materials combining advantages of both battery and electrochemical capacitor device technologies. Carbon nanotubes (CNT) have gained widespread attention for a range of electrochemical energy storage and conversion device applications given their unique properties, including electrical conductivity, high surface area, and chemical and mechanical stability. Lee and colleagues investigated a new class of electrodes for energy storage devices facilitated by Li+ reactions with functional surface groups on the MWNT electrode structure. Combining this with a high surface area transition metal oxide lithium storage material at the opposing negative electrode, the authors have demonstrated unprecedented performance for energy storage device electrodes potentially impacting a range of hybrid applications. |
Reviewed by Jeff Morse, PhD., National Nanomanufacturing Network
- Lee SW, Kim BS, Chen S, Shao-Horn Y, and Hammond PT. 2009. Layer-by-layer Assembly of All Carbon Nanotube Ultrathin Films for Electrochemical Applications. JACS 131(2):671-679. DOI: 10.1021/ja807059k.
- Lee SW, Yabuuchi N, Gallant BN, Chen S, Kim BS, Hammond PT, and Shao-Horn Y. 2010.High-powered Lithium Batteries from Functionalized Carbon Nanotube Electrodes. Nature Nanotechnology 5:531-537. DOI: 10.1038/nnano.2010.116.
Emerging hybrid energy storage applications require characteristics bridging the attributes and performance gaps between batteries and electrochemical capacitors. Applications such as electric vehicles and renewable energy sources require the development of materials combining advantages of both device technologies, including high energy density provided by Faradaic reaction in batteries, and high rate power delivery provided by surface redox reactions in electrochemical capacitors. Thus a power source providing rapid charge and discharge with superior gravimetric energy density remains essential to the long-term success of these applications. Research efforts have considered this challenge from both directions, including increasing the power density of lithium-ion (L+) batteries by reducing the lithium diffusion time through decreased thickness of the lithium storage materials. In this approach, nanostructured electrode materials still exhibit lower power density in comparison to electrochemical capacitors. On the other hand, nanostructured carbon and transition metal oxide materials have been investigated as a means to increase the energy density and charge storage capability of capacitor technologies.
In order to demonstrate the functional design of specific thin film architectures, precise control of CNT film thickness, density, and adhesion properties is necessary. One such approach developed by the Hammond group at MIT, referred to as layer-by-layer (LBL) assembly, effectively assembles thin films of densely woven multiwall nanotube (MWNT) structures via electrostatic attraction of functionalized MWNT’s from highly dispersed aqueous solutions. Recently, Lee et.al. extended this technique through investigation of additive-free MWNT assemblies with stable pseudo-capacitive functional groups. Combining this functional LBL-MWNT electrode with a lithiated Li4Ti5O12 (LTO) negative electrode, the authors demonstrated electrode structures having enhanced energy and power density in comparison to batteries and supercapacitors. The authors theorized that the high gravimetric energy density is a result of Faradaic reactions between the lithium ions and the functional groups on the MWNT surface.
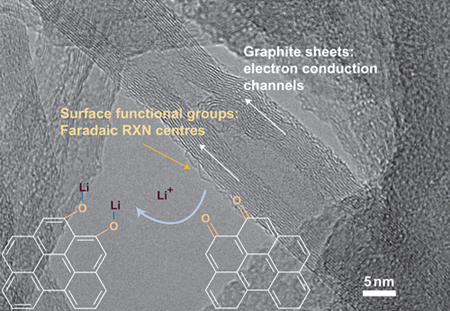
Image reproduced by permission from MacMillan Publishers: Lee SW, Yabuuchi N, Gallant BN, Chen S, Kim BS, Hammond PT, and Shao-Horn Y. 2010.High-powered Lithium Batteries from Functionalized Carbon Nanotube Electrodes. Nature Nanotechnology 5:531-537. DOI: 10.1038/nnano.2010.116, copyright 2010.
This work is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported.
