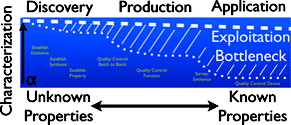
A prevalent challenge for progress in nanotechnology is characterization [1, 2]. Characterization, the measurement of various physicochemical properties of materials, is crucial for the evolution of nanotechnology from rudimentary nanomaterials and devices to those that are precision-engineered, mass producible, and safe. Each of the main sectors of nanotechnology – research, manufacturing, and regulation – needs systematic characterization in order to maximize knowledge and control of nanomaterials. Due to a number of factors, many of the nanomaterials that have been synthesized thus far are poorly defined, which can lead to false generalizations about performance and toxicity. The advancement of nanotechnology depends upon a coordinated effort made by researchers, manufacturers, regulators, and funding agencies to improve characterization techniques and practices so that well-defined and reproducible nanomaterials are studied and manufactured. An important consequence of thoroughly characterized materials will be increased public awareness, acceptance and use of nanotechnology.
In order for a nanomaterial to be well-characterized, what are the properties that must be measured? Because of the challenges inherent in creating and working with materials at the nanoscale, the answer to this question is complex and elusive. Numerous academic, industrial, and governmental groups have been working for several years to identify the properties that would constitute a minimum characterization set for nanomaterials, but nationally- or internationally-recognized standards have not been established yet. However, a recent study reviewed 28 lists of properties that were suggested by the aforementioned groups for inclusion in a minimum characterization set. The study found that several properties were common to most of the lists: size, shape, surface area, core composition, surface chemistry, surface charge, and agglomeration or aggregation state [2]. These properties could serve as an unofficial minimum characterization set for nanomaterials until formal standards are in place. Along with establishing standards, the advancement of nanotechnology will be hastened by developing instruments that are more sensitive and capable of simultaneously measuring multiple properties of a single sample.
Among the nanomaterial properties mentioned above, the importance of surface chemistry, which may be defined as the composition, structure, and reactivity of the surface of a material, has been significantly underemphasized, especially for unsupported nanomaterials such as particles in suspension or powder form [2]. As a particle’s size decreases, its surface becomes increasingly dominant compared to core atoms, and therefore, surface composition, structure, and reactivity become the governing properties of nanoparticles [3]. Furthermore, it is the outer surface of a particle that is exposed to the surrounding environment and interacts with other materials; the interface is where the action is. Studies are emerging that demonstrate the importance of surface chemistry on nanomaterial behavior and underscore the need for researchers and manufacturers to rigorously characterize surfaces. For example, in a toxicity study, Qui and colleagues [4] synthesized gold nanorods with four different aspect ratios and three different surface coatings and found that although both aspect ratio and surface chemistry affect cellular internalization, only the surface chemistry played a role in cytotoxicity.
When inadequately characterized, nanomaterials are often referred to with overly general terms, leaving out important property details that would hone a material’s identity. Thus far, the bulk of published studies concerning nanotoxicity or environmental fate are vague in their descriptions of the nanomaterials used in investigations, often only reporting the core material, size, and surface charge of the particles. In platform and poster presentations at a 2013 nanotoxicity workshop held at a prestigious nanoresearch center, generalizations ran rampant, with statements such as “gold nanoparticles caused inflammation” or “quantum dots were shown to be toxic.” While particle diameters were provided, surface chemistry and other important properties were often neglected. As is the case for many types of nanomaterials, gold nanoparticles and quantum dots can vary widely in their core and shell compositions, size, shape, and surface chemistry, allowing for a vast range of formulations, each yielding unique particle behavior. Thus, in order to push nanotechnology to a more advanced level, vague statements such as "5-nm quantum dots are toxic” need to be replaced with more descriptive labels when articulating research hypotheses or reporting experimental results. For example, “5-nm cadmium selenide quantum dots stabilized with mercapto-acetic acid and conjugated to the antibody herceptin are toxic to HeLa cells” [5].
Insufficient characterization coupled with inadequate reporting of important material properties has the potential to undeservedly tarnish the image of a broad range of nanomaterials, particularly when experimental results indicating a harmful effect might apply only to a specific formulation. A negative image of nanomaterials in the eyes of consumers, regulators, or potential funding bodies could hinder the acceptance and use of nanotechnology and, consequently, create significant barriers to the growth of nanomanufacturing. Avoiding this undesirable outcome requires well-defined materials whose behavior can be explained or associated with measured physicochemical properties. As Lord Kelvin suggested over one hundred years ago, to measure is to know, and if something is not measured, it cannot be improved. The more nanomaterials are characterized, the better scientists and manufacturers can demonstrate to the public that nanomaterials are understood, controllable, and useful. Richman and Hutchison [6] eloquently summarize the issue: “For the moment, the public belief is that ‘nano is good.’ Maintaining the public trust benefits both research and business. However, without strong, clear evidence, the public will make up their own mind based upon social rather than physical data. We need methods of characterization that are fast, facile, and formal to preserve the public trust and to maximize the future of nanotechnology.”
References:
[1] Cressey, D. Tiny Traits Cause Big Headaches. Nature 2010, 467(7313), 264-265. DOI: 10.1038/467264b
[2] Baer, D.R.; Engelhard, M.H.; Johnson, G.E.; et al. Surface Characterization of Nanomaterials and Nanoparticles: Important Needs and Challenging Opportunities. J. Vac. Sci. Technol., A 2013, 31(5), 050820. DOI: 10.1116/1.4818423
[3] Grainger, D.W.; Castner, D.G. Nanobiomaterials and Nanoanalysis: Opportunities for Improving the Science to Benefit Biomedical Technologies. Adv. Mater. 2008, 20(5), 867-877. DOI: 10.1002/adma.200701760
[4] Qui, Y.; Liu, Y.; Wang, L.; et al. Surface Chemistry and Aspect Ratio Mediated Cellular Uptake of Au Nanorods. Biomaterials 2010, 31(30), 7606-7619. DOI: 10.1016/j.biomaterials.2010.06.051
[5] Tsoi, K.M.; Dai, Q.; Alman, B.A.; et al. Are Quantum Dots Toxic? Exploring the Discrepency Between Cell Culture and Animal Studies. Acc. Chem. Res.2013, 43(3), 662-671. DOI: 10.1021/ar300040z
[6] Richman, E.K.; Hutchison, J.E. The Nanomaterial Characterization Bottleneck. ACS Nano 2009, 3(9), 2441-2446. DOI: 10.1021/nn901112p
Image reprinted with permission from The Nanomaterial Characterization Bottleneck, Erik K. Richman and James E. Hutchison, ACS Nano 2009 3 (9), 2441-2446 Copyright 2009 American Chemical Society
