| Zhu, et. al., report on the use of “nanodome” solar cells to simultaneously improve optical absorption and enhance surface cleanliness. The resulting structures have power conversion efficiencies of 5.9%. |
Reviewed by Fred Sharifi, National Institute of Standards and Technology
There has been almost continuous research on improving photovoltaic (PV) efficiency through optical designs that enhance collection efficiency. A fair amount of this research has centered on three‐dimensional structures fabricated through etch (e.g. grooves in Si) or conformal growth that result in improved collection efficiency through multiple reflections. Zhu, et. al., have extended this approach to the nanoscale, allowing for enhanced optics leading to improved absorption, superhydrophobic functionality of the surface, and, perhaps critically, enhanced charge collection in a potentially promising PV absorber material which lacks extended electronic states.
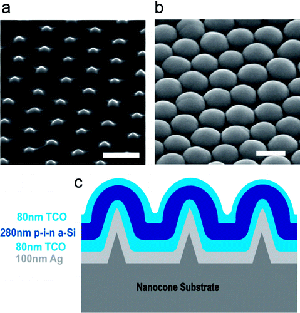
The authors describe the fabrication process in detail. The initial template is formed by using monodispersed SiO2 particles assembled into closed pack layers on a quartz substrate. This template is subsequently plasma etched using standard oxide etch chemistry, resulting in “nanocones” on the substrate. This is followed by the fabrication of the p‐i‐n “nanodomes” through PECVD deposition of a‐Si:H. It is not apparent what developments would be required to make this fabrication technique scalable, resulting in large‐scale PV cells based on these structures.
The authors’ data demonstrates a 25% improvement, resulting in cells with 5.9% conversion efficiency. This improvement is mostly the result of an efficient and relatively flat absorbance between 400 and 800 nm in these nanostructured cells, with 94% absorption. Further, the design is such that this high absorption efficiency persists for radiation incident over a large angular range. These improved optics result in a short circuit current that is near the theoretical value imposed by the band gap of the material. The benefits of the shorter charge collection paths in these nanostructured cells on charge collection efficiency, and how it is manifested in power conversion efficiency will require more work to quantify completely.
Another interesting advantage of these structures is that they have superhydrophobic properties, and may potentially suffer less from performance degradation over time due to dirt accumulation on the surface of the PV cell than conventional cells. The authors demonstrate enhanced “self‐cleaning” properties through surface chemical treatment that introduce hydrophobic molecules.
Image reproduced with permission from Zhu J, et.al. 2009. Nanodome Solar Cells with Efficient Light Management and Self Cleaning. Nano Letters Article ASAP 9 November 2009. DOI: 10.1021/nl9034237. Copyright 2009 American Chemical Society.
