ISO/TR 13121:2011 offers guidance on the information needed to make sound risk evaluations and risk management decisions, as well as how to manage in the face of incomplete or uncertain information by using reasonable assumptions and appropriate risk management practices. Further, ISO/TR 13121:2011 includes methods to update assumptions, decisions, and practices as new information becomes available, and on how to communicate information and decisions to stakeholders.
ISO/TR 13121:2011 suggests methods organizations can use to be transparent and accountable in how they manage nanomaterials. It describes a process of organizing, documenting, and communicating what information organizations have about nanomaterials.
ISO website:ISO/TR 13121:2011
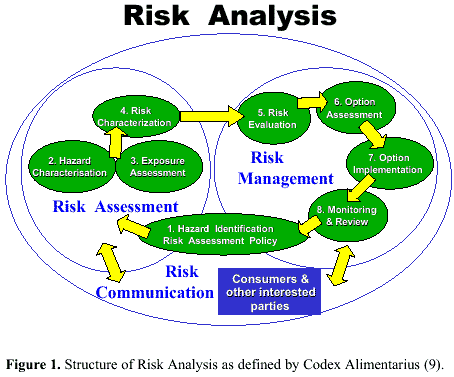
Image source: Reij MW and Van Schothorst M. Critical notes on microbiological risk assessment of food. Braz. J. Microbiol. 2000; 31(1). doi: 10.1590/S1517-83822000000100002
