| Gao, et. al., report a versatile, one-step process technology for producing f-CNTs based on nitrene chemistry. |
Reviewed by Jeff Morse, Ph.D., National Nanomanufacturing Network
The functionalization of carbon nanotubes (CNTs) is important for improving CNT dispersion characteristics and controlling the chemical and physical properties of either single wall nanotubes (SWNTs) or multi wall nanotubes (MWNTs). These materials are excellent candidates for use in a range of biotechnology, nanotechnology, and catalytic process applications. While low cost, industrial production of pristine MWNTs and SWNTs is presently available, schemes for efficient, cost effective functionalization and dispersion remain under investigation with the goal of achieving a breakthrough technology for scaled production.
While several diverse strategies have been developed, the predominant method to functionalize CNTs incorporates the nitric acid oxidation method to introduce carboxylic acid groups onto the surface of the CNT. If desired, conversion to other functional groups can be achieved through subsequent chemical reactions. While this technique is a powerful method, clearly improving CNT dispersion, several disadvantages exist as a result of the acid treatment process. These include the use of corrosive acids for the processes which are both expensive and environmentally unfriendly, the low yield of the reaction which consumes large portions of the quality CNTs thereby diminishing the economy of scale, the defects generated during acid treatment that impair the CNTs’ properties and morphology, and the limited control of specific functional groups attached to the CNTs’ surface. In this respect, scalable synthesis of functionalized CNTs using this method faces barriers associated with too many process steps, process complexity, environmental concerns, cost, and versatility of subsequent functionalization and surface reactions.
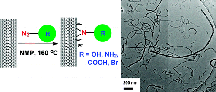
The benefits of this methodology for scaled production include environmentally- friendly chemistries and processes, synthesis of azide materials and compounds under fairly mild process conditions, high specificity for desired functional group chemistry, and no severe damage to the CNTs during the chemical process. Additionally, the f-CNTs were subsequently chemically modified to synthesize surface initiated polymerization of various types of monomers, along with the generation of Pt/CNT nanohybrid structures. The potential and benefit of this approach may lead to the scalable synthesis of f-CNTs, along with the ability to control the specific functional groups positioned on the pristine nanostructures.
By developing a simple one-step approach, improvements in characterization and understanding of specific chemical bonds and associated processes may additionally be provided. Further investigations are warranted to expand the available functional group chemistries and subsequent chemical modification methods.
Image reproduced with permission from Gao, C., He, H., Zhou, L., Zheng, X., and Zhang, Y., "Scalable Functional Group Engineering of Carbon Nanotubes by One-Step Nitrene Chemistry." Chem Mater 21 (2) 2009 360 - 370. Copyright 2008, American Chemical Society.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported.
