With rising levels of atmospheric carbon dioxide and indicators for global climate change increasingly apparent, the search has intensified for more sustainable and renewable alternative energy sources. Photovoltaic (PV) energy has been of interest for the last 40 years; however, the cost of Si-based solar panels is still not cost-effective for widespread usage. In addition to the overall cost, consumer adoption of this technology has been slow due to the unattractive aesthetics of traditional solar panels. Building integrated photovoltaics (BIPVs) such as Dow PowerhouseTM Solar Shingles have reached the market, but have not yet been widely adopted. In order for PV technology to be widely accepted, it will need to be seamlessly incorporated into existing infrastructures such as building and automotive materials, and be available in a variety of colors.
Among the various types of emerging PV technologies such as dye-sensitized solar cells (DSSCs), organic cells, and quantum dots (QDs), perovskite solar cells represent one of the most promising sectors. In a span of only 5 years, the efficiency of perovskite PVs has increased from ca. 3% to a current level in excess of 20%. This efficiency is now comparable to that of crystalline Si and semiconductor thin films (e.g., copper indium gallium selenide (CIGS), CdTe) that have been in development since the 1970’s. Perovskite solar technology utilizes a hybrid inorganic-organic halide perovskite (e.g., CH3NH3MI3 where M = Sn, Pb) in combination with n- and p-type charge collection layers.
In a promising step toward aesthetically attractive solar panels, Zhang and coworkers describe the fabrication of perovskite photovoltaic devices that are color-tunable. In order to maximize the refractive index contrast among the layers in the device, nanoparticulate films of porous SiO2 (50% porosity) and dense TiO2 (4% porosity) were deposited by simple spin-coating. The combination of the photonic properties of the oxide nanoparticle films and absorptive properties of the overlying perovskite material was used to fine-tune the observed color range from orange to blue. Interestingly, the strategy employed by these researchers may be considered as a biomimetic approach, since beetles and butterflies also employ reflective and absorptive layers that yield a characteristic and often tunable color. Many gemstones such as opals also utilize the photonic crystal effect to give rise to iridescent colors. The use of a photonic phenomenon for color generation in this multilayered PV device is preferred rather than using dyes or pigments, since the latter would likely fade over time.
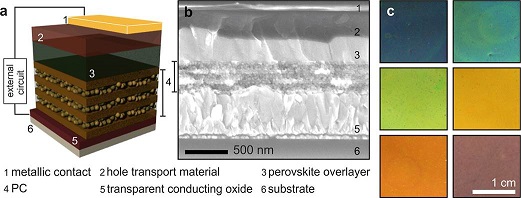
Reference: Zhang W, Anaya M, Lozano G, Calvo ME, Johnston MB, Míguez H, Snaith HJ. Highly Efficient Perovskite Solar Cells with Tunable Structural Color. Nano Letters. 2015; 15 (3): 1698-1702 doi: 10.1021/nl504349z
Image reprinted with permission from American Chemical Society.
