| Typical graphene composite synthesis methods generate films with random orientation and positioning of the graphene nanostructures. However, there remains significant interest in exploring pathways to synthesize graphene superstructures with well-ordered and aligned architectures. In particular, vertically aligned graphene layer arrays (VAGLAs) could potentially enable a range of unique properties such as anisotropic thermal and electrical conductivity. Guo et al. demonstrate a process utilizing a range of CLC precursor molecules to synthesize vertically aligned graphene layer arrays with heights ranging from 50-800 nm with full 2D order. |
Reviewed by Jeff Morse, Ph.D., National Nanomanufacturing Network
- Guo F, Mukhopadhyay A, Sheldon BW, and Hurt RH. 2010. Vertically Aligned Graphene Layer Arrays from Chromonic Liquid Crystal Precursors. Advanced Materials 23(4):508-513. doi:10.1002/adma.201003158.
Due to a range of unique and superior properties, including high thermal and electrical conductivity, interest in graphene has increased dramatically in recent years. Progress in synthesis methods have led to prospects for graphene-based transistors, sensors, and large-area, transparent electrodes. Typical synthesis methods include depositing graphene monolayers on flat substrates and surfaces, which are subsequently integrated into device structures through normal semiconductor fabrication processes, or exfoliating graphite into graphene flakes, which are then dispersed into composite films having unique properties or relocated on other substrates for further process integration. While graphene dispersions offer some routes to recast graphene composite films—and potentially manipulate their alignment, location, and ordering—the resulting films typically have random orientation and positioning of the graphene nanostructures. There remains significant interest in exploring pathways to synthesize graphene superstructures with well-ordered and aligned architectures. In particular, vertically aligned graphene layer arrays (VAGLAs) could potentially enable a range of unique properties such as anisotropic thermal and electrical conductivity. Additionally, the availability of a high concentration of exposed edge-sites on the top surface would enable effective functionalization at high densities to realize superhydrophobic/philic coatings, high redox-activity electrodes, or rapid charge/discharge cyling for lithium ion batteries.
∏-stacking within aqueous solutions, behaving as supramolecular rods, which then order into nematic liquid crystal phases via rod self-avoidance at high concentrations. Thus the isotropic-nematic (I-N) phase transition occurs from entropic effects, wherein the packing and orientation entropies are offset at high rod densities in concentrated solution. The result is parallel orientation of supramolecular rods on substrate surface producing sets of vertically aligned disks that may be converted to graphene layers while retaning vertical alignment and ordering.
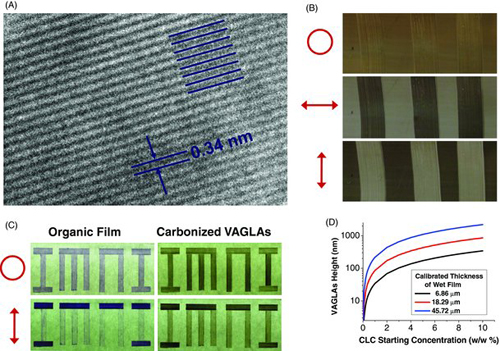
The authors investigated the synthesis of VAGLAs utilizing five separate molecular precursors. which were sulfonated polyaromatic dyes having hydrophilic periphery and hydrophobic core, observing the I-N transition as a function of polyaromatic molecular concentration, and drying conditions. The resulting supramolecular rod length could be predicted from the estimated molecule size, material density, and concentration in solution. Typical rods synthesized were 14-80 nm in length with 40-240 vertically aligned disks at nominal spacing of 0.34 nm.
In order to fabricate uniform VAGLA heights the authors found that the precursor solutions must be applied with uniform film thickness in the presence of shear or elongation flow in order to align the supramolecular rods. Incorporating the Mayer-bar application method, shear and elongational flows at the interface of the substrate align the rod-shaped aggregates in the direction that the bar is drawn. Therefore the disks are aligned perpendicular to both the substrate and the drawing direction. Thus, by controlling the shear rate in the gap to maintain laminar flow of the solution during application, ordered rod alignment patterns are produced. The resulting CLC films were then dried at room temperature creating solid crystal films and subsequently carbonized by heating in nitrogen at 700°C for 30 minutes. During the carbonization step, the orientation of the disk-like molecules in the rod structure is stabilized through edge-to-edge polymerization reactions in which neighboring molecular disks cross-link, forming vertical carbon sheets exhibiting the unique graphene assembly pattern and crystal structure. Of the five chromonic precursors studied, the authors were able to generate VAGLAs having two-dimensional order from each, but found that the beginning precursor concentration needed to be greater than the I-N transition concentration otherwise multiple coating passes were necessary to achieve stable anisotropy. Additionally, it was found that VAGLA height could be controlled through appropriate selection of precursor concentration and Mayer-bar wire dimension. Thus a process has been demonstrated utilizing a range of CLC precursor molecules to synthesize vertically aligned graphene layer arrays with heights ranging from 50-800 nm with full 2D order.
Image reprodced from Guo F, Mukhopadhyay A, Sheldon BW, and Hurt RH. 2010. Vertically Aligned Graphene Layer Arrays from Chromonic Liquid Crystal Precursors. Advanced Materials 23(4):508-513. doi:10.1002/adma.201003158. Permission pending.
This work is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported.
