| A straightforward, scalable approach has been demonstrated to create a more efficient 3-D nanostructured electrode material for electrochemical applications. The method incorporates an innovative biotemplating approach with a top-down conformal coating process to realize an operationally beneficial nanonetwork structure. |
Reviewed by Jeff Morse, Ph.D, National Nanomanufacturing Network
- Kim SW, Han TH, Kim J, Gwon H, Moon HS, Kang SW, Kim SO, Kang K. 2009. Fabrication and Electrochemical Characterization of TiO2 Three-Dimensional Nanonetwork Based on Peptide Assembly. ACS Nano 3(5):1085-1090 . DOI:10.1021/nn900062q.
Nanostructured materials have gained significant attention for energy storage and conversion devices owing to the large surface areas that are possible. In the case of large surface area nanostructured electrodes for rechargeable Li-ion batteries, the interaction between electrode and electrolyte provides benefits for battery operation. Specifically, nanoscale electrodes can reduce the Li ion diffusion length, which leads to enhanced rate capability, and can accommodate more of the strain induced during Li insertion/extraction cycles that typically lead to limited cycle stability. Another aspect to consider in optimizing 3-D nanostructures for electrode materials is the ability for the electrolyte to penetrate the inner diameter of the structure. In some configurations, small diameters limit penetration of even moderate amounts of electrolyte, effectively eliminating this portion of the electrode surface area.
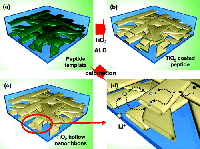
Electrochemical testing of the 3-D titania nanonetwork electrodes demonstrated superior rate capability, particularly at higher current densities in comparison to titania nanopowders. The biotemplated nanonetwork exhibited a 5-fold increase in specific capacity under the rate of charge/discharge as compared to 100 nm diameter titania nanopowder. Additionally, the templated nanonetwork exhibited exceptional cyclability, retaining >80% of the initial capacity after 200 charge/discharge cycles. The low average capacity fade and high Coulomb efficiency per cycle is a stark contrast to the poor behavior of Titania nanopowders. The authors attribute the performance improvements to the high specific area and short Li ion diffusion path offered via the nanonetwork structure. As compared to the nanopowders, the hollow nanoribbon network offers benefits of facile electronic conduction through the ordered network, along with Li diffusion from both the outside and inner hollow space of the tube.
A straightforward, scalable approach has been demonstrated to create a more efficient 3-D nanostructured electrode material for electrochemical applications. The method incorporates an innovative biotemplating approach with a top-down conformal coating process to realize an operationally beneficial nanonetwork structure.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported.
