
Through advances made in material characterization and nanoscale analysis, it is now known that medieval glaziers colored their glass with gold nanoparticles, although they were unaware of the nanoparticulate structure of which their colorants were composed. With modern knowledge of how nanostructures can often give materials unique optical or catalytic properties, a team in Australia and China has developed a novel gold nanoparticle catalyst that converts polluting volatile organic compounds (VOCs) such as carbon monoxide and formaldehyde into non-toxic carbon dioxide. Gold nanoparticles are known to oxidize VOCs, but the authors of this new research observed that the oxidation can occur even at ambient temperatures when irradiated with visible light, due to the same light-absorbing quality that gives the nanoparticles their glass-coloring properties.
Application of external heat to activate the catalyst, as in previous efforts, requires heating the catalyst support material as well as the catalyst itself. The researchers propose that visible light absorption by the gold nanoparticles produces heat in the vicinity of the nanoparticle, thus activating the catalyst. Since visible light absorption at ambient temperature causes heating only in the vicinity near the catalyst, potentially large energy savings could result from this technology. As shown in the figure, they observed that formaldehyde conversion by the gold nanoparticles increased to ~64% by increasing the intensity of the light incident on the gold nanoparticle catalyst. 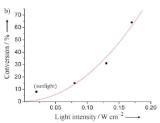
Applications of this research include treatment of indoor air pollution from furnishings that off-gas VOCs, or treatment of waste process gases. For example, the authors have suggested that medieval gold nanoparticle-containing glass was capable of converting VOCs to carbon dioxide, and similarly, catalytic surfaces could be incorporated into modern day buildings for improved indoor air quality. Additionally, the researchers indicate that their work with light-driven catalysis could potentially be extended to drive other reactions, such as conventional gold catalysis, at ambient temperatures for significant energy savings.
Several other research teams are also investigating gold nanoparticles for VOC treatment, including a team from NIST, Lehigh University and Cardiff University, who have recently published work in Science showing that the extent of carbon monoxide oxidation by gold nanoparticles is strongly dependent on the size of the nanoparticles. The work presented in Science was performed at ambient temperatures without the use of light irradiation.
Additional Supporting Material:
Other links:
- V. Aguilar-Guerrero, B.C. Gates, “Genesis of a highly active cerium oxide-supported gold catalyst for CO oxidation,” Chem. Comm. 2007, p. 3210.
- Casey, Scott. "Church windows cleanse the soul - and the air," Brisbane Times, August 24, 2008.
- NEW SCIENCE STUDY STUDYING GOLD NP’S AND SIZE EFFECTS
- Herzing, A.A., Kiely, C.J., Carley, A.F., Landon, P., Hutchings, G.J. “Identification of Active Gold Nanoclusters on Iron Oxide Supports for CO Oxidation” Science 2008, 321, 1331.
Images from Visible-Light-Driven Oxidation of Organic Contaminants in Air with Gold Nanoparticle Catalysts on Oxide Supports. Angewandte Chemie Int'l Ed. 2008 47 5353. Copyright Wiley-VCH Verlag Gmbh& Co. Reproduced with permission.
